A novel stimulator device to treat gait deficits in multiple sclerosis (MS) patients with mild to moderate symptoms earned a nod from the FDA Friday.
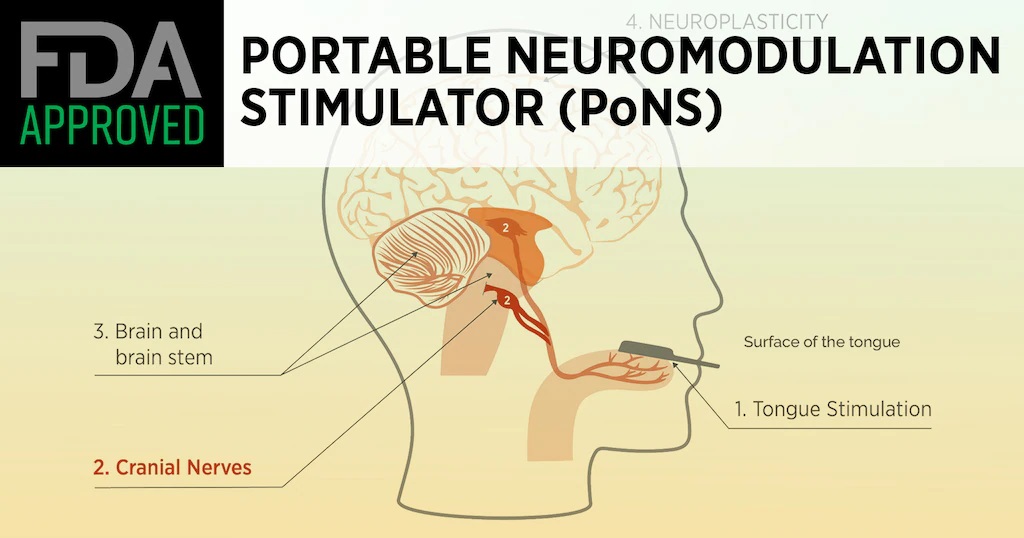
The device, known as the Portable Neuromodulation Stimulator (PoNS), generates electrical pulses on the tongue to stimulate trigeminal and facial nerves to treat motor deficits. To be available by prescription, it is to be used only as part of a supervised therapeutic exercise program in MS patients 22 and older, the agency said. The FDA authorized the device through its “de novo” premarket review pathway for new devices posing low to moderate risks for adverse effects.
“MS is one of the most common neurological diseases in young adults. Today’s authorization offers a valuable new aid in physical therapy and increases the value of additional therapies for those who live with MS on a daily basis,” Christopher Loftus, MD, acting director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health, said in a statement.
Most people with MS experience their first symptoms between the ages of 20 and 40 and the disease occurs more frequently in women than in men. MS can cause a variety of symptoms, including problems with walking and balance.
The PoNS device delivers mild neuromuscular electrical stimulation to the dorsal surface of the patient’s tongue. A control unit, worn around the neck, sends signals to a mouthpiece the patient holds lightly in place with their lips and teeth. A therapist can view usage data later to identify “potential areas of missed or shortened sessions,” the FDA noted. To read this article in its entirety click this link: New Device for MS Gait
